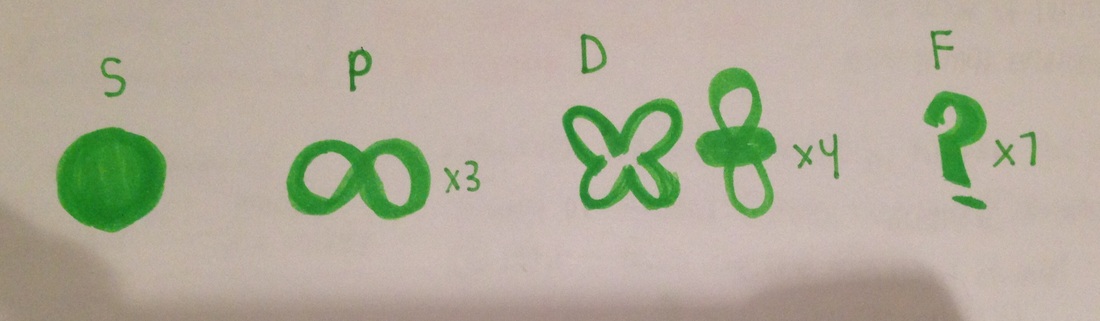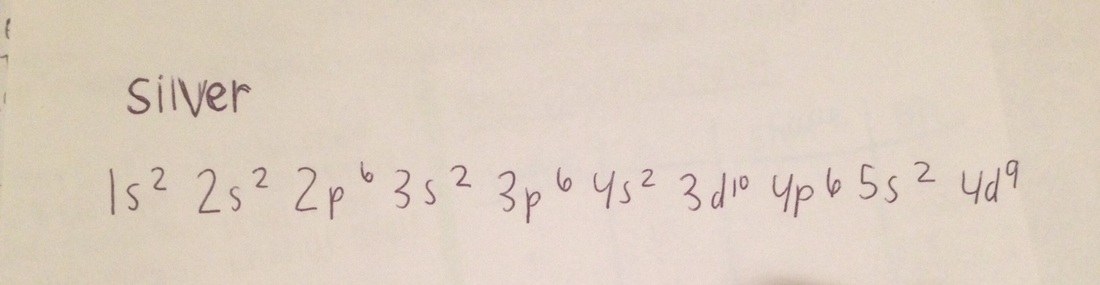chapter 11: modern atomic theory
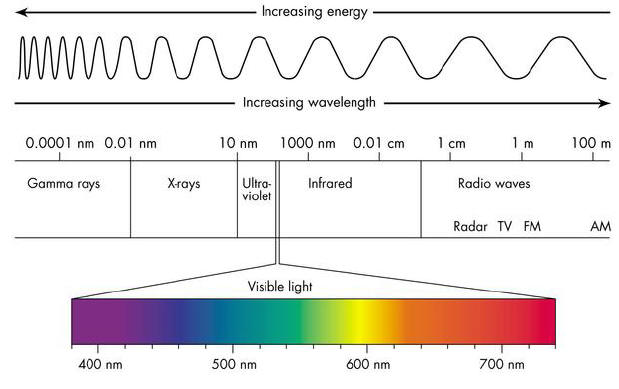
Electromagnetic Radiation is a form of energy that exhibits wave-like behavior as it travels through space and time
- Radio Waves - communication
- Microwaves - used to cook
- Infrared - “heat waves”
- Visible Light - detected by your eyes
- Ultraviolet - causes sunburns; study DNA
- X-rays - penetrates tissue
- Gamma Rays - most energetic
Energy Levels of Atoms
- Electrons can be found in the ground state. When energy is added to the atom electron can jump to higher energy levels and becomes known as the excited state. These electron can relax down to a lower energy level and release the energy as light (photon).
Quantized energy levels vs. continuous energy levels
- certain discrete energy levels that will always emit photons
- certain discrete energy levels that will always emit photons
Wave Mechanical Model of the Atom
- Wave Mechanic can suggest the probability of the location of the electron
- Probability map (orbitals) describe electrons in the lowest possible energy states
- The model gives no information about when electrons occupy points in space or how they move
- 4 kinds of orbitals:
- S, P, D, F
- S = sphere
- P= infinity /hour glass (3 of these)
- D= butterfly wings (4 of these)
- F= 7 shapes (unknown)
- Pauli exclusion principal states that an atomic orbital can hold a maximum of two electrons and those two electrons must have opposite spins
Electron Configuration/Orbital Diagram Examples
1. Name the electron configuration of Silver
1. Name the electron configuration of Silver
2. Draw the Orbital diagram of silver
3. write the shortcut for calcium electron configuration